Home / Diazomethane (CH2N2)
Carboxylic Acid Derivatives
Diazomethane (CH2N2)
Last updated: May 25th, 2023 |
Reactions Of Diazomethane (CH2N2) And Their Mechanisms
- Diazomethane is a useful (albeit highly toxic and potentially explosive) reagent for the conversion of carboxylic acids to methyl esters.
- It can also be used for the one-carbon extension (“homologation”) of carboxylic acids via the Arndt-Eistert synthesis.
- The excellent leaving group (N2) makes it a potential source of carbenes, which can be used for cyclopropanation of alkenes.
- Diazomethane (CH2N2) is the simplest diazo compound. A gas at room temperature (boiling point -23°C) it is generally prepared and used as a dilute solution with specialized equipment (such as flame-polished joints) that minimizes the chance of explosion. A related (and safer) reagent, trimethylsilyldiazomethane (TMSCHN2) is increasingly used in place of diazomethane.
Table of Contents
-
- The Lewis Structure of Diazomethane (CH2N2)
- Conversion of Carboxylic Acids To Methyl Esters With Diazomethane
- Mechanism of Methyl Ester Formation
- “Homologation” of Carboxylic Acids Using Diazomethane in the Arndt-Eistert Synthesis
- Mechanism of the Arndt-Eistert Synthesis (and Wolff Rearrangement)
- Cyclopropanations With Diazomethane
- Summary
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. The Lewis Structure of Diazomethane (CH2N2)
Diazomethane is the simplest diazo compound, consisting of a methylene carbon (CH2) bonded to dinitrogen (N2 ).
Just for fun, before we go any further, see if you can draw a proper Lewis structure for diazomethane, CH2N2.
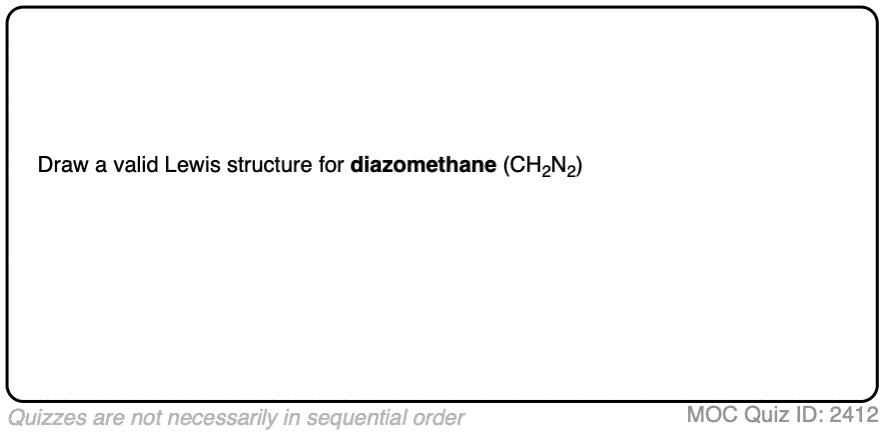 Click to Flip
Click to Flip
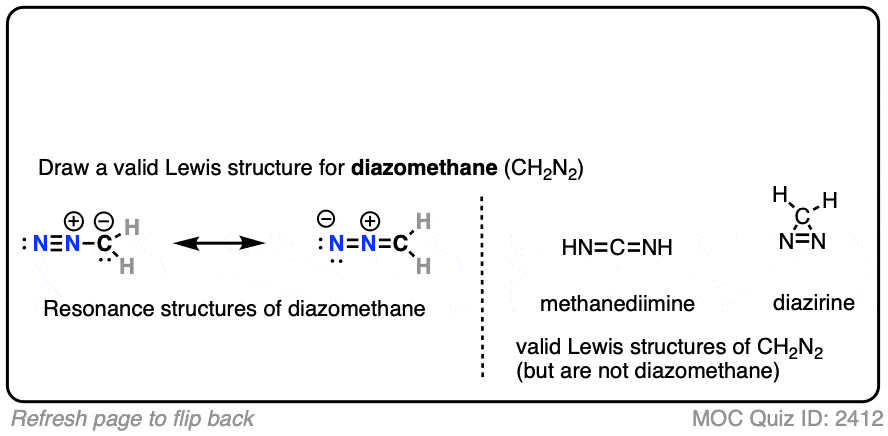
Note the two important resonance forms of diazomethane. The key resonance form for our purposes is the one on the left (on the flip side of the quiz), where carbon bears a lone pair and a negative formal charge. The other two compounds shown on the right are also valid Lewis structures for CH2N2 but are not diazomethane. [Note 1]
This provides an important clue to the potential reactivity of diazomethane; it is nucleophilic on carbon, and is also a reasonably good base. (The pKa of its conjugate acid is about 10, similar to that of NH4(+), so diazomethane is about as basic as NH3. )
The other clue to the chemical behavior of diazomethane is found in that nitrogen-nitrogen triple bond, which is the ultimate reason why diazomethane is the source of so many laboratory horror stories.
(For an example, see this article: Diazomethane – How Not To Do It)
Good leaving groups are weak bases – and dinitrogen (N2) is one of the weakest bases of all. As you might well be aware, every nitrogen atoms’ goal in life is to regain the freedom it once had as a molecule of dinitrogen (N2) in the earth’s atmosphere – and in diazomethane, this can be attained merely through breaking of a single carbon-nitrogen bond.
This makes diazomethane very frisky, and highly unstable towards things we would ordinarily classify as innocuous, like ground-glass joints and metal syringe tips.
Furthermore, if jostled, shocked, heated, or even treated with strong enough ultraviolet light, diazomethane will rapidly and violently give off nitrogen gas.
For these reasons [and also that it is a yellow gas at room temperature (boiling point –23 °C)] it is generally prepared and handled as a dilute solution in a solvent such as ether using special equipment (i.e. flame-polished pipettes, and no ground glass joints). [Note 2]
And oh, by the way, it is also extremely toxic. (See Note 3 for some details on handling diazomethane).
So why bother using it at all? (Because it’s the best! Note 4 )
2. Conversion of Carboxylic Acids To Methyl Esters With Diazomethane
Well, carboxylic acids can be made into methyl esters by treating them with the reagent diazomethane, CH2N2.
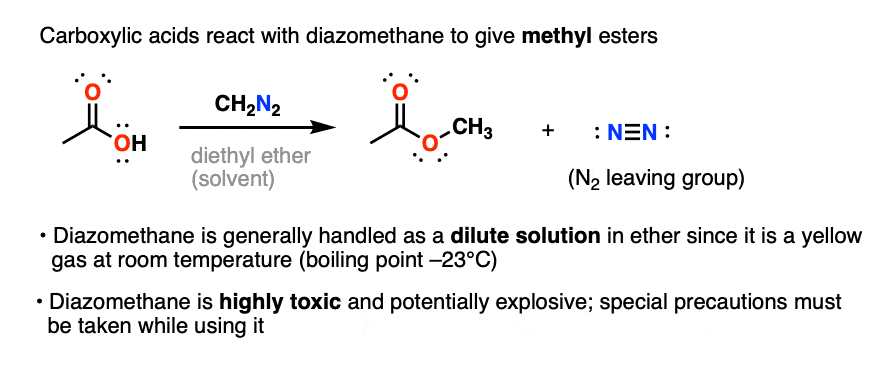
This is a very mild way of converting carboxylic acids into esters. Due to the aforementioned toxicity and explosive hazards of diazomethane, it’s usually done on small scale.
An alternative (and much safer) reagent, trimethylsilyldiazomethane (TMSCHN2) is increasingly used in place of CH2N2. (Note 5)
3. Carboxylic Acids To Methyl Esters With Diazomethane – Mechanism
The conversion of carboxylic acids to methyl esters is a two-step process.
In the first step, diazomethane, a weak base, is protonated on carbon to give H3C-N2(+). This is an example of a diazonium compound, which you may have encountered if you’ve covered the Sandmeyer and related reactions.
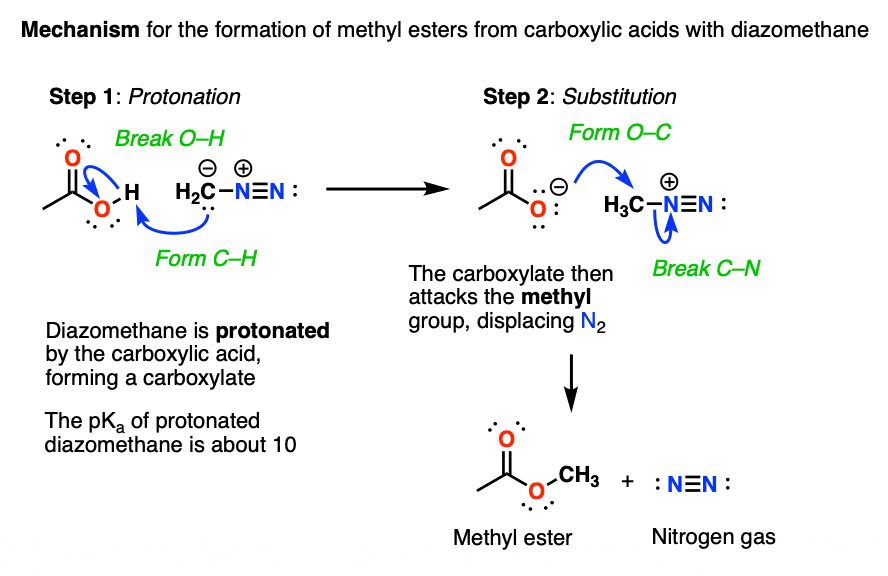
Like most diazonium compounds, CH3-N2(+) doesn’t stick around for long. It quickly recombines with the resulting carboxylate anion in an SN2 reaction to give the methyl ester and liberates the molecule of N2 to join its brethren in the atmosphere.
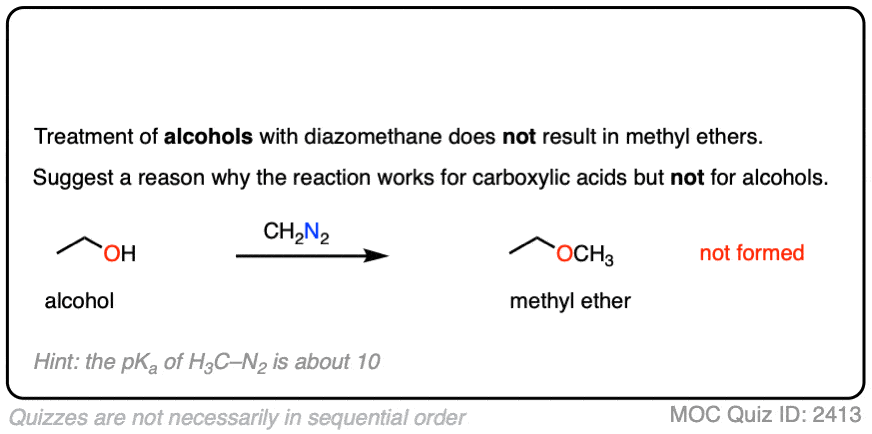
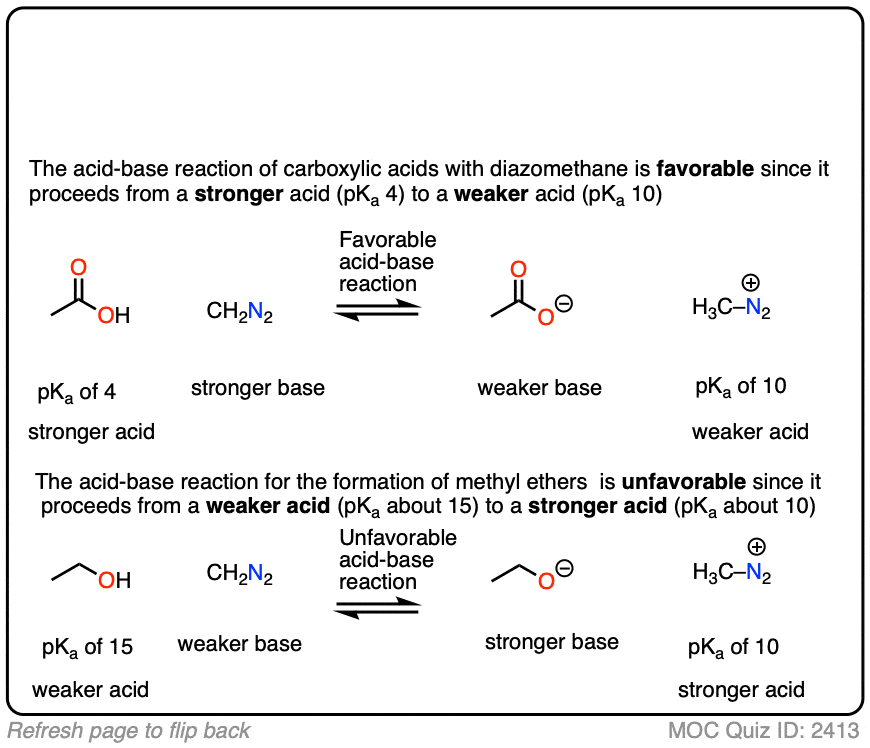
Interestingly, while diazomethane reacts readily with carboxylic acids, it does not undergo reactions with alkyl alcohols such as ethanol and propanol.
Can you figure out why? (Hint: the pKa of H3C-N2(+) is about 10). (Note 6)
 Click to Flip
Click to Flip
4. “Homologation” Of Carboxylic Acids With Diazomethane Using the “Arndt-Eistert Synthesis”
Diazomethane is also used for a process called the homologation of carboxylic acids, which is a fancy term for “making them one carbon longer”.
In this way, one could start with, say, acetic acid (CH3COOH) and convert it to propanoic acid (CH3CH2COOH).
A handy trick! (if you enjoy the smell of vomit – but I digress)
The source of the “extra” carbon is the CH2 of diazomethane. This process was developed about a hundred years ago and is often referred to the Arndt-Eistert synthesis.
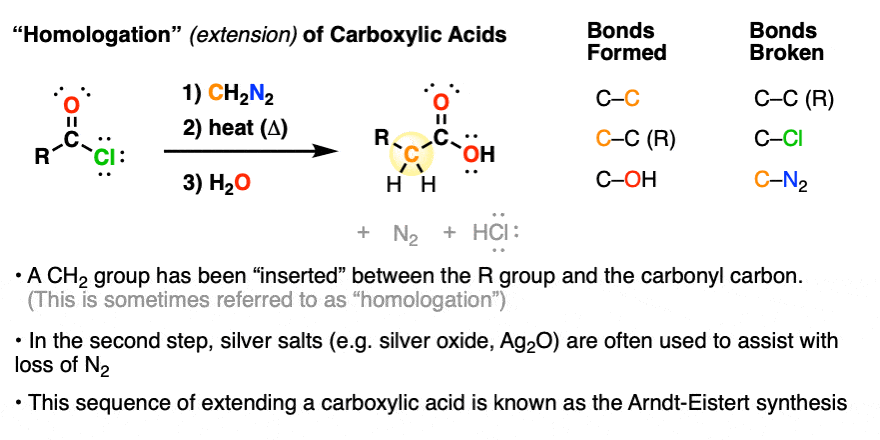
- The process starts with an acid chloride (which can be made from the carboxylic acid through treatment with thionyl chloride, oxalyl chloride, PCl5 or similar reagents). (See article: Thionyl Chloride)
- Then, diazomethane is added (usually at least two equivalents, although modern procedures require just one).
- Heating (or more commonly, addition of a silver salt such as Ag2O) results in loss of nitrogen gas accompanied by a skeletal rearrangement known as the Wolff Rearrangement, giving an intermediate product known as a ketene.
- Addition of water to the ketene gives the carboxylic acid.
5. Mechanism of the Arndt-Eistert Synthesis (and Wolff Rearrangement)
There are three main phases in the Arndt-Eistert synthesis.
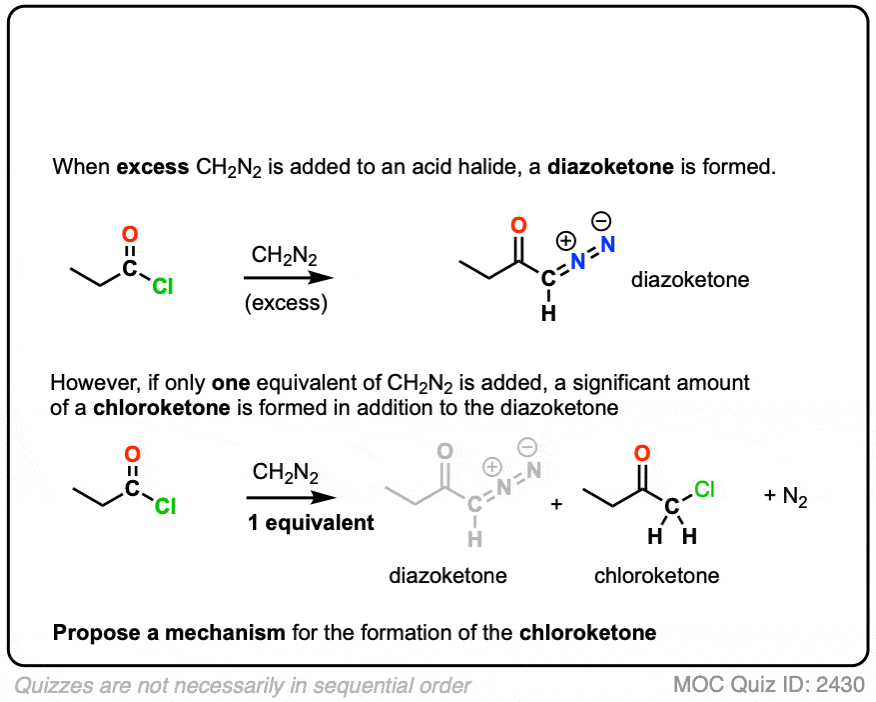
The first is reaction of diazomethane with the acid halide to give a new species known as a diazoketone.
- Acid halides readily undergo nucleophilic acyl substitution to give various carboxylic acid deriviates. (See article: Nucleophilic Acyl Substitution)
- Diazomethane is a reasonably good nucleophile due to the presence of the lone pair on carbon.
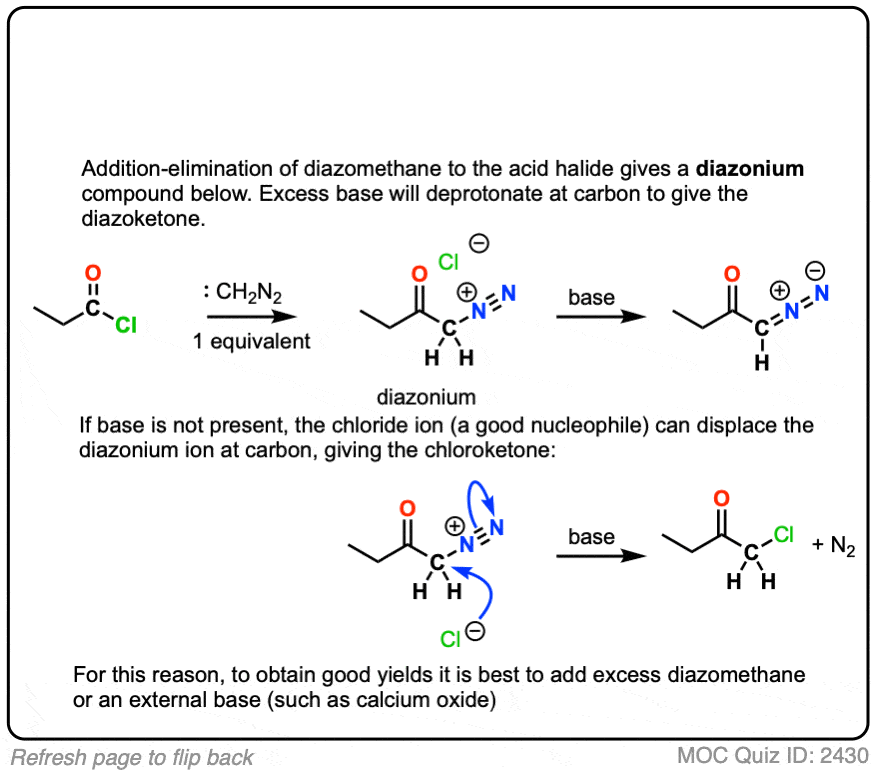
So the first two steps are a relatively straightforward addition-elimination process that give a new diazonium compound.
We’re not quite done.
Being adjacent to the carbonyl (and to N2) the alpha carbon is reasonably acidic and can be deprotonated by excess diazomethane (or an external base such as calcium oxide, see here) to give an enolate.
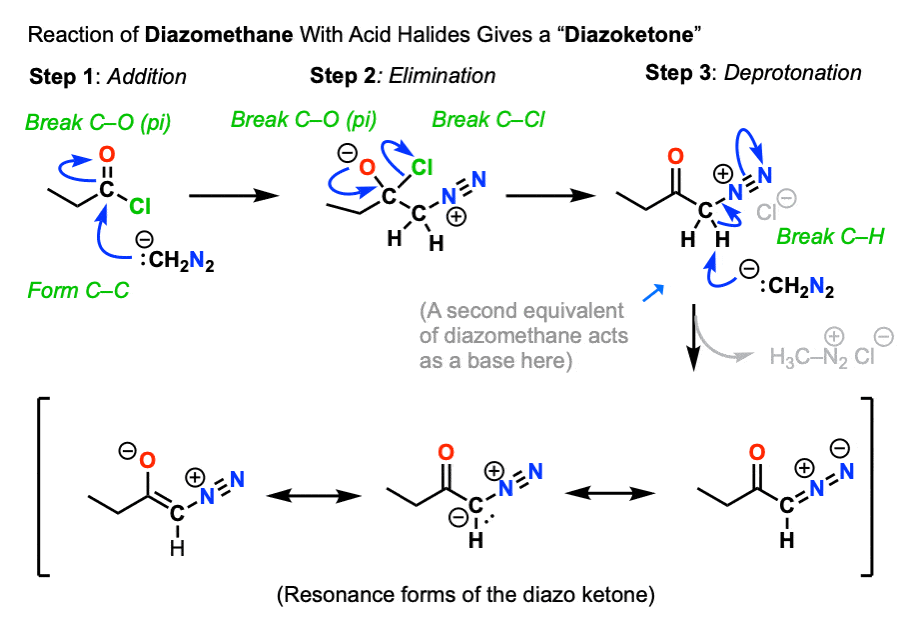
The enolate is just one of three main resonance forms of this molecule, which is often referred to as a “diazoketone”.
Keep an eye on that “enolate” resonance form (far left), however.
The next step is where things get a little weird.
If you start by drawing that resonance form on the far left, you can imagine a process where
- the C-O pi bond forms, while
- the C-C bond breaks, and migrates to the adjacent carbon (similar to 1,2-hydride and 1,2-alkyl shifts), and
- the C-N bond breaks, liberating N2
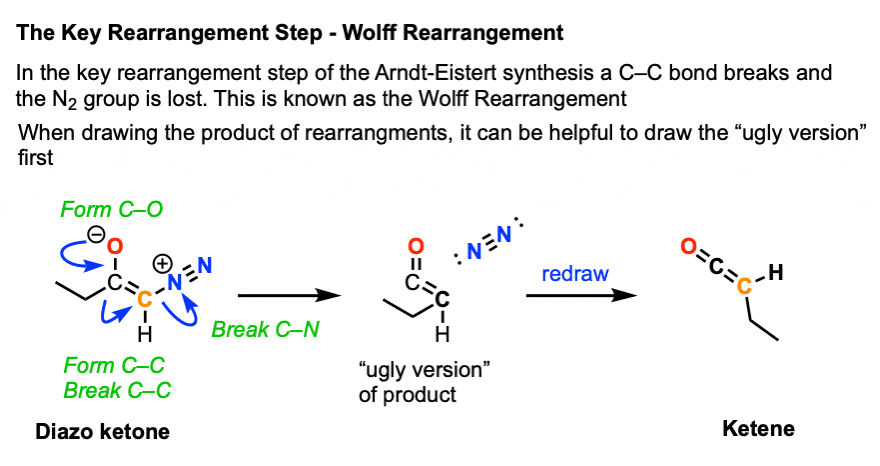
(This rearrangement bears a lot of similarities to the Curtius and Hofmann rearrangements – see article)
It can be helpful to draw an ugly looking version of this product first to get the connectivity right.
The product of this reaction is known as a ketene, which is something of an exotic species for introductory organic chemistry.
Ketenes are excellent electrophiles. The C=O carbon of ketenes undergo reactions with various nucleophiles to give carboxylic acid derivatives.
They will even react with weak nucleophiles like water, giving a carboxylic acid.
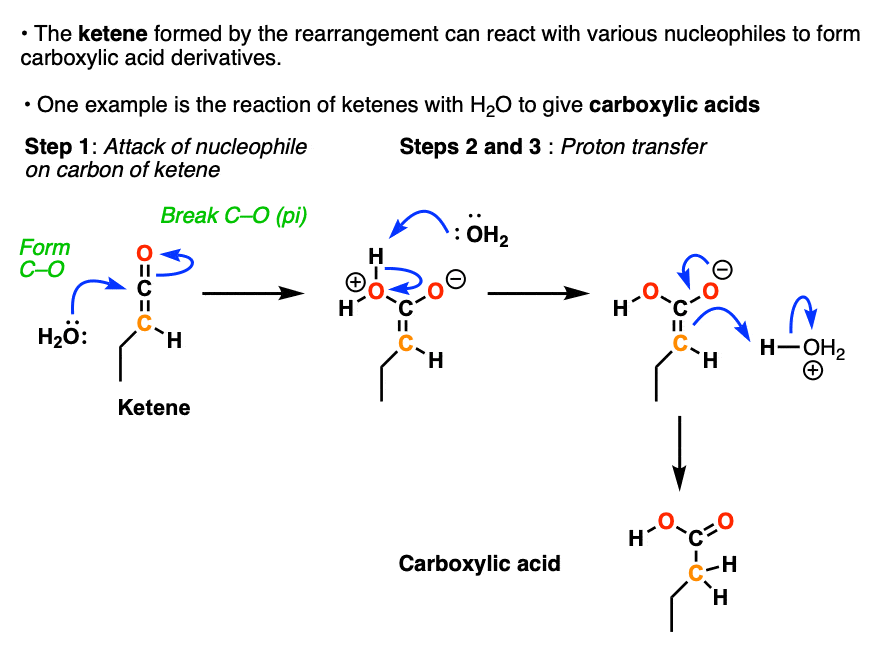
Ketenes will similarly react with alcohols to give esters and amines to give amides.
6. Cyclopropanation with Diazomethane
One final application of diazomethane in introductory organic chemistry is as a source of carbenes.
Carbenes are divalent carbon atoms (i.e. bonded to two atoms) and possess two non-bonded electrons. Like carbocations, they have six valence electrons and are electron-deficient species.
When heated, treated with light, or even in the presence of certain metals, diazomethane will lose N2 to give “methylene carbene”, :CH2
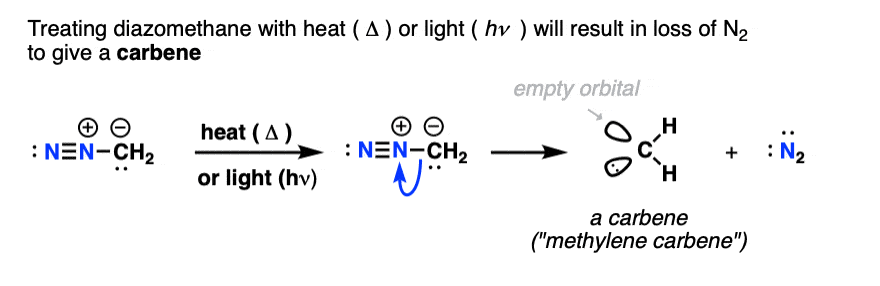
In the presence of alkenes, carbenes will form cyclopropanes.
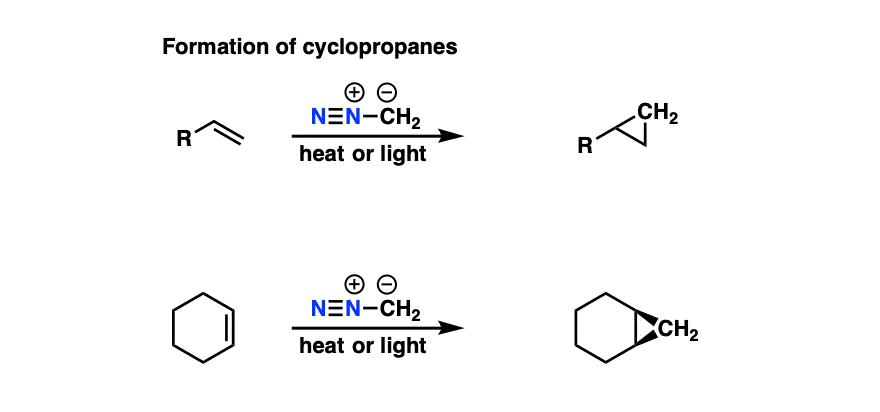
Rather than heat or light (which can be unselective), palladium and copper catalysts are often used for these reactions. (For a representative example see here. )
7. Summary
Diazomethane has its uses. In introductory organic chemistry, these are primarily:
- Formation of methyl esters from carboxylic acids
- In the formation of diazo ketones, and subsequent Wolff rearrangement
- Cyclopropanation
It is, to say the least, highly unlikely that an undergraduate laboratory will be using diazomethane for any reason, as it is extremely toxic and potentially explosive. In the event that you are in the position of having to carry out a procedure with CH2N2, it should be done with the utmost care and only with proper training and equipment.
Notes
Related Articles
- The Hofmann and Curtius Rearrangements
- Nucleophilic Acyl Substitution (With Negatively Charged Nucleophiles)
- Thionyl Chloride (SOCl2)
- Formation of cyclopropanes from alkenes using methylene carbene (:CH2) (MOC Membership)
- Wolff Rearrangement (MOC Membership)
- How to Use a pKa Table
- Basicity of Amines And pKaH
- Thionyl Chloride (SOCl2)
Note 1. The two other compounds are valid Lewis structures but are not diazomethane.
- HN=C=NH is the simplest example of a carbodiimide, the most prominent example of which is dicyclohexylcyclodiimide (DCC).
- The cyclic molecule is called diazirine. Like diazomethane, it can also lose N2 to give methylene carbene. Although it looks extremely unstable, it has been synthesized – see here (J. Am. Chem. Soc. 1962, 84, 1063).
Note 2. A common precursor for preparing diazomethane is through the basic decomposition of p-tolylsulfonylmethylnitrosamide, which is sold by Aldrich under the trade name, DIAZALD®. They also sell special kits for the preparation of diazomethane that have flame-polished glass joints. A representative procedure for making diazomethane is here.
Note 3: Tips on working with diazomethane. Man, there are so many. Here’s one: don’t ever, ever work with diazomethane unless you have been trained by someone who has worked with it themselves. Although generally never used neat (it boils at –23 °C) and typically handled as a dilute solution in ether, diazomethane has a nasty tendency to explode when in the presence of high-surface-area materials (such as ground-glass joints, or, God forbid, metal syringes). If dispensing with a pipet, it’s crucial to flame-polish the edges for safety. Work behind a blast shield.
This article has useful tips on safe handling of diazomethane.
Furthermore, diazomethane is acutely toxic: people have died from inhaling it. And if it doesn’t kill you in the short term, there’s always the long term. See, diazomethane, being an alkylating agent, has a tendency to react with (i.e. alkylate) your DNA, leading to base-pair mismatches, mutations, and – eventually – cancer.
Check out the amount of red ink on this preparation from Organic Syntheses. That should tell you a lot.
Note 4: Why bother using diazomethane at all? Because it’s the best. The formation of methyl esters from the reaction of carboxylic acids with diazomethane is one of my favorite reactions. You make a solution of your carboxylic acid in ether, dispense the bright yellow diazomethane solution by pipet – slowly – until the yellow color persists, wait about 5 minutes to be sure that all the N2 has bubbled off, kill the excess diazomethane with a few drops of acetic acid, and concentrate. No column required. Easiest reaction ever.
Note 5. What about using, say, diazoethane to make ethyl esters, or diazopropane to make propyl esters? It can be done – see here – the problem is getting a good yield of the diazoalkanes (30% is typical for older procedures). One more recent paper has better yields. (See reference 10)
Note 6. The reaction works better with more acidic alcohols like phenols (pKa 10). The reaction can be made to work with alkyl alcohols if a Lewis acid like BF3 is added.

Quiz Yourself!
 Click to Flip
Click to Flip
 Click to Flip
Click to Flip
(Advanced) References and Further Reading
For some riveting, mouth-agape horror stories of diazomethane in the lab, check out this thread from In the Pipeline : Diazomethane: How Not To Do It.
Diazomethane is a reagent commonly used in organic chemistry for a variety of transformations. It is too reactive to be sold as a pure compound; it is generated from precursors in solution and used immediately.
- Ueber Diazomethan
V. Pechmann
Chem. Ber. 1894, 27 (2), 1888-1891
DOI: 10.1002/cber.189402702141 - Ueber Diazomethan
v. Pechmann
Chem. Ber. 1895, 28 (1), 855-861
DOI: 10.1002/cber.189502801189
The first two papers by Hans von Pechmann on the synthesis and discovery of diazomethane. - DIAZOMETHANE
J. de Boer and H. J. Backer
Org. Synth. 1956, 36, 16
DOI: 10.15227/orgsyn.036.0016
This procedure, the base-catalyzed decomposition of p-tolylsulfonylmethylnitrosamide, is the most common method for preparing diazomethane today. The diazomethane is generated as an ethereal solution and should be used quickly. Aldrich sells kits for the preparation of diazomethane based on this, and the precursor is commercially available and is commonly called ‘DIAZALD®’. - Improved Arndt−Eistert Synthesis of α-Diazoketones Requiring Minimal Diazomethane in the Presence of Calcium Oxide as Acid Scavenger
Vittorio Pace, Guido Verniest, Josep-Vicent Sinisterra, Andrés R. Alcántara, and Norbert De KimpeThe Journal of Organic Chemistry 2010 75 (16), 5760-5763
DOI: 10.1021/jo101105g
This 2010 procedure uses calcium oxide as a base to scavenge HCl during the formation of diazoketones. - DIDEUTERIODIAZOMETHANE
G. Gassman and W. J. Greenlee
Org. Synth. 1973, 53, 38
DOI: 10.15227/orgsyn.053.0038
Note 1 in this procedure has detailed notes on safety regarding the use of diazomethane. Teflon stirbars should be used, metal should be avoided, the solution kept away from light, and glassware with ground-glass joints should not be used. - Iron-Catalyzed Cyclopropanation in 6 M KOH with in Situ Generation of Diazomethane
Bill Morandi and Erick M. Carreira
Science 23 Mar 2012: Vol. 335, Issue 6075, pp. 1471-1474
DOI: 1126/science.1218781
This paper describes a ‘one-pot’ cyclopropanation of styrenes and related compounds without the prior isolation of diazomethane, thereby making this chemistry safer and more practical. - Trimethylsilyl-substituted diazoalkanes : I. Trimethylsilyldiazomethane
Dietmar Seyferth, Horst Menzel, Alan W. Dow, Thomas C. Flood
Journal of Organometallic Chemistry 1972, 44 (2), 279-290
DOI: 1016/S0022-328X(00)82916-2
Trimethylsilyldiazomethane (aka ‘TMS-diazomethane’) is a much easier to handle alternative to diazomethane. It is commercially available and can be used for much of the same reactions that diazomethane undergoes. - Rapid and convenient isolation and methyl esterification of water-soluble acids using diazomethane
J. Eisenbraun, R. N. Morris, and G. Adolphen
Journal of Chemical Education 1970, 47 (10), 710
DOI: 10.1021/ed047p710
This is a short note that describes a modification to the usual esterification procedure with diazomethane to facilitate the esterification of water-soluble carboxylic acids. - Mechanism of Methyl Esterification of Carboxylic Acids by Trimethylsilyldiazomethane
Erik Kühnel, David D. P. Laffan, Guy C. Lloyd‐Jones, Teresa Martínez del Campo, Ian R. Shepperson, Jennifer L. Slaughter
Angew. Chem. Int. Ed. 2007, 46 (37), 7075-7078
DOI: 10.1002/anie.200702131
The esterification of carboxylic acids is a common reaction of diazoalkanes, especially diazomethane. TMS-diazomethane can also be used for this reaction as a safer alternative to diazomethane - Catalytic Homologation of Cycloalkanones with Substituted Diazomethanes. Mild and Efficient Single-Step Access to α-Tertiary and α-Quaternary Carbonyl Compounds
David C. Moebius and Jason S. KingsburyJournal of the American Chemical Society 2009 131 (3), 878-879
DOI: 10.1021/ja809220j
This paper contains procedures for making a large variety of substituted diazomethane derivatives. It also has extremely useful notes on safe handling and preparation of diazomethane and its derivatives.
00 General Chemistry Review
01 Bonding, Structure, and Resonance
- How Do We Know Methane (CH4) Is Tetrahedral?
- Hybrid Orbitals and Hybridization
- How To Determine Hybridization: A Shortcut
- Orbital Hybridization And Bond Strengths
- Sigma bonds come in six varieties: Pi bonds come in one
- A Key Skill: How to Calculate Formal Charge
- The Four Intermolecular Forces and How They Affect Boiling Points
- 3 Trends That Affect Boiling Points
- How To Use Electronegativity To Determine Electron Density (and why NOT to trust formal charge)
- Introduction to Resonance
- How To Use Curved Arrows To Interchange Resonance Forms
- Evaluating Resonance Forms (1) - The Rule of Least Charges
- How To Find The Best Resonance Structure By Applying Electronegativity
- Evaluating Resonance Structures With Negative Charges
- Evaluating Resonance Structures With Positive Charge
- Exploring Resonance: Pi-Donation
- Exploring Resonance: Pi-acceptors
- In Summary: Evaluating Resonance Structures
- Drawing Resonance Structures: 3 Common Mistakes To Avoid
- How to apply electronegativity and resonance to understand reactivity
- Bond Hybridization Practice
- Structure and Bonding Practice Quizzes
- Resonance Structures Practice
02 Acid Base Reactions
- Introduction to Acid-Base Reactions
- Acid Base Reactions In Organic Chemistry
- The Stronger The Acid, The Weaker The Conjugate Base
- Walkthrough of Acid-Base Reactions (3) - Acidity Trends
- Five Key Factors That Influence Acidity
- Acid-Base Reactions: Introducing Ka and pKa
- How to Use a pKa Table
- The pKa Table Is Your Friend
- A Handy Rule of Thumb for Acid-Base Reactions
- Acid Base Reactions Are Fast
- pKa Values Span 60 Orders Of Magnitude
- How Protonation and Deprotonation Affect Reactivity
- Acid Base Practice Problems
03 Alkanes and Nomenclature
- Meet the (Most Important) Functional Groups
- Condensed Formulas: Deciphering What the Brackets Mean
- Hidden Hydrogens, Hidden Lone Pairs, Hidden Counterions
- Don't Be Futyl, Learn The Butyls
- Primary, Secondary, Tertiary, Quaternary In Organic Chemistry
- Branching, and Its Affect On Melting and Boiling Points
- The Many, Many Ways of Drawing Butane
- Wedge And Dash Convention For Tetrahedral Carbon
- Common Mistakes in Organic Chemistry: Pentavalent Carbon
- Table of Functional Group Priorities for Nomenclature
- Summary Sheet - Alkane Nomenclature
- Organic Chemistry IUPAC Nomenclature Demystified With A Simple Puzzle Piece Approach
- Boiling Point Quizzes
- Organic Chemistry Nomenclature Quizzes
04 Conformations and Cycloalkanes
- Staggered vs Eclipsed Conformations of Ethane
- Conformational Isomers of Propane
- Newman Projection of Butane (and Gauche Conformation)
- Introduction to Cycloalkanes
- Geometric Isomers In Small Rings: Cis And Trans Cycloalkanes
- Calculation of Ring Strain In Cycloalkanes
- Cycloalkanes - Ring Strain In Cyclopropane And Cyclobutane
- Cyclohexane Conformations
- Cyclohexane Chair Conformation: An Aerial Tour
- How To Draw The Cyclohexane Chair Conformation
- The Cyclohexane Chair Flip
- The Cyclohexane Chair Flip - Energy Diagram
- Substituted Cyclohexanes - Axial vs Equatorial
- Ranking The Bulkiness Of Substituents On Cyclohexanes: "A-Values"
- Cyclohexane Chair Conformation Stability: Which One Is Lower Energy?
- Fused Rings - Cis-Decalin and Trans-Decalin
- Naming Bicyclic Compounds - Fused, Bridged, and Spiro
- Bredt's Rule (And Summary of Cycloalkanes)
- Newman Projection Practice
- Cycloalkanes Practice Problems
05 A Primer On Organic Reactions
- The Most Important Question To Ask When Learning a New Reaction
- Curved Arrows (for reactions)
- Nucleophiles and Electrophiles
- The Three Classes of Nucleophiles
- Nucleophilicity vs. Basicity
- What Makes A Good Nucleophile?
- What Makes A Good Leaving Group?
- 3 Factors That Stabilize Carbocations
- Equilibrium and Energy Relationships
- 7 Factors that stabilize negative charge in organic chemistry
- 7 Factors That Stabilize Positive Charge in Organic Chemistry
- What's a Transition State?
- Hammond's Postulate
- Learning Organic Chemistry Reactions: A Checklist (PDF)
- Introduction to Oxidative Cleavage Reactions
06 Free Radical Reactions
- Bond Dissociation Energies = Homolytic Cleavage
- Free Radical Reactions
- 3 Factors That Stabilize Free Radicals
- What Factors Destabilize Free Radicals?
- Bond Strengths And Radical Stability
- Free Radical Initiation: Why Is "Light" Or "Heat" Required?
- Initiation, Propagation, Termination
- Monochlorination Products Of Propane, Pentane, And Other Alkanes
- Selectivity In Free Radical Reactions
- Selectivity in Free Radical Reactions: Bromination vs. Chlorination
- Halogenation At Tiffany's
- Allylic Bromination
- Bonus Topic: Allylic Rearrangements
- In Summary: Free Radicals
- Synthesis (2) - Reactions of Alkanes
- Free Radicals Practice Quizzes
07 Stereochemistry and Chirality
- Types of Isomers: Constitutional Isomers, Stereoisomers, Enantiomers, and Diastereomers
- How To Draw The Enantiomer Of A Chiral Molecule
- How To Draw A Bond Rotation
- Introduction to Assigning (R) and (S): The Cahn-Ingold-Prelog Rules
- Assigning Cahn-Ingold-Prelog (CIP) Priorities (2) - The Method of Dots
- Enantiomers vs Diastereomers vs The Same? Two Methods For Solving Problems
- Assigning R/S To Newman Projections (And Converting Newman To Line Diagrams)
- How To Determine R and S Configurations On A Fischer Projection
- The Meso Trap
- Optical Rotation, Optical Activity, and Specific Rotation
- Optical Purity and Enantiomeric Excess
- What's a Racemic Mixture?
- Chiral Allenes And Chiral Axes
- Stereochemistry Practice Problems and Quizzes
08 Substitution Reactions
- Nucleophilic Substitution Reactions - Introduction
- Two Types of Nucleophilic Substitution Reactions
- The SN2 Mechanism
- Why the SN2 Reaction Is Powerful
- The SN1 Mechanism
- The Conjugate Acid Is A Better Leaving Group
- Comparing the SN1 and SN2 Reactions
- Polar Protic? Polar Aprotic? Nonpolar? All About Solvents
- Steric Hindrance is Like a Fat Goalie
- Common Blind Spot: Intramolecular Reactions
- Substitution Practice - SN1
- Substitution Practice - SN2
09 Elimination Reactions
- Elimination Reactions (1): Introduction And The Key Pattern
- Elimination Reactions (2): The Zaitsev Rule
- Elimination Reactions Are Favored By Heat
- Two Elimination Reaction Patterns
- The E1 Reaction
- The E2 Mechanism
- E1 vs E2: Comparing the E1 and E2 Reactions
- Antiperiplanar Relationships: The E2 Reaction and Cyclohexane Rings
- Bulky Bases in Elimination Reactions
- Comparing the E1 vs SN1 Reactions
- Elimination (E1) Reactions With Rearrangements
- E1cB - Elimination (Unimolecular) Conjugate Base
- Elimination (E1) Practice Problems And Solutions
- Elimination (E2) Practice Problems and Solutions
10 Rearrangements
11 SN1/SN2/E1/E2 Decision
- Identifying Where Substitution and Elimination Reactions Happen
- Deciding SN1/SN2/E1/E2 (1) - The Substrate
- Deciding SN1/SN2/E1/E2 (2) - The Nucleophile/Base
- SN1 vs E1 and SN2 vs E2 : The Temperature
- Deciding SN1/SN2/E1/E2 - The Solvent
- Wrapup: The Key Factors For Determining SN1/SN2/E1/E2
- Alkyl Halide Reaction Map And Summary
- SN1 SN2 E1 E2 Practice Problems
12 Alkene Reactions
- E and Z Notation For Alkenes (+ Cis/Trans)
- Alkene Stability
- Alkene Addition Reactions: "Regioselectivity" and "Stereoselectivity" (Syn/Anti)
- Stereoselective and Stereospecific Reactions
- Hydrohalogenation of Alkenes and Markovnikov's Rule
- Hydration of Alkenes With Aqueous Acid
- Rearrangements in Alkene Addition Reactions
- Halogenation of Alkenes and Halohydrin Formation
- Oxymercuration Demercuration of Alkenes
- Hydroboration Oxidation of Alkenes
- m-CPBA (meta-chloroperoxybenzoic acid)
- OsO4 (Osmium Tetroxide) for Dihydroxylation of Alkenes
- Palladium on Carbon (Pd/C) for Catalytic Hydrogenation of Alkenes
- Cyclopropanation of Alkenes
- A Fourth Alkene Addition Pattern - Free Radical Addition
- Alkene Reactions: Ozonolysis
- Summary: Three Key Families Of Alkene Reaction Mechanisms
- Synthesis (4) - Alkene Reaction Map, Including Alkyl Halide Reactions
- Alkene Reactions Practice Problems
13 Alkyne Reactions
- Acetylides from Alkynes, And Substitution Reactions of Acetylides
- Partial Reduction of Alkynes With Lindlar's Catalyst
- Partial Reduction of Alkynes With Na/NH3 To Obtain Trans Alkenes
- Alkyne Hydroboration With "R2BH"
- Hydration and Oxymercuration of Alkynes
- Hydrohalogenation of Alkynes
- Alkyne Halogenation: Bromination, Chlorination, and Iodination of Alkynes
- Alkyne Reactions - The "Concerted" Pathway
- Alkenes To Alkynes Via Halogenation And Elimination Reactions
- Alkynes Are A Blank Canvas
- Synthesis (5) - Reactions of Alkynes
- Alkyne Reactions Practice Problems With Answers
14 Alcohols, Epoxides and Ethers
- Alcohols - Nomenclature and Properties
- Alcohols Can Act As Acids Or Bases (And Why It Matters)
- Alcohols - Acidity and Basicity
- The Williamson Ether Synthesis
- Ethers From Alkenes, Tertiary Alkyl Halides and Alkoxymercuration
- Alcohols To Ethers via Acid Catalysis
- Cleavage Of Ethers With Acid
- Epoxides - The Outlier Of The Ether Family
- Opening of Epoxides With Acid
- Epoxide Ring Opening With Base
- Making Alkyl Halides From Alcohols
- Tosylates And Mesylates
- PBr3 and SOCl2
- Elimination Reactions of Alcohols
- Elimination of Alcohols To Alkenes With POCl3
- Alcohol Oxidation: "Strong" and "Weak" Oxidants
- Demystifying The Mechanisms of Alcohol Oxidations
- Protecting Groups For Alcohols
- Thiols And Thioethers
- Calculating the oxidation state of a carbon
- Oxidation and Reduction in Organic Chemistry
- Oxidation Ladders
- SOCl2 Mechanism For Alcohols To Alkyl Halides: SN2 versus SNi
- Alcohol Reactions Roadmap (PDF)
- Alcohol Reaction Practice Problems
- Epoxide Reaction Quizzes
- Oxidation and Reduction Practice Quizzes
15 Organometallics
- What's An Organometallic?
- Formation of Grignard and Organolithium Reagents
- Organometallics Are Strong Bases
- Reactions of Grignard Reagents
- Protecting Groups In Grignard Reactions
- Synthesis Problems Involving Grignard Reagents
- Grignard Reactions And Synthesis (2)
- Organocuprates (Gilman Reagents): How They're Made
- Gilman Reagents (Organocuprates): What They're Used For
- The Heck, Suzuki, and Olefin Metathesis Reactions (And Why They Don't Belong In Most Introductory Organic Chemistry Courses)
- Reaction Map: Reactions of Organometallics
- Grignard Practice Problems
16 Spectroscopy
- Degrees of Unsaturation (or IHD, Index of Hydrogen Deficiency)
- Conjugation And Color (+ How Bleach Works)
- Introduction To UV-Vis Spectroscopy
- UV-Vis Spectroscopy: Absorbance of Carbonyls
- UV-Vis Spectroscopy: Practice Questions
- Bond Vibrations, Infrared Spectroscopy, and the "Ball and Spring" Model
- Infrared Spectroscopy: A Quick Primer On Interpreting Spectra
- IR Spectroscopy: 4 Practice Problems
- 1H NMR: How Many Signals?
- Homotopic, Enantiotopic, Diastereotopic
- Diastereotopic Protons in 1H NMR Spectroscopy: Examples
- 13-C NMR - How Many Signals
- Liquid Gold: Pheromones In Doe Urine
- Natural Product Isolation (1) - Extraction
- Natural Product Isolation (2) - Purification Techniques, An Overview
- Structure Determination Case Study: Deer Tarsal Gland Pheromone
17 Dienes and MO Theory
- What To Expect In Organic Chemistry 2
- Are these molecules conjugated?
- Conjugation And Resonance In Organic Chemistry
- Bonding And Antibonding Pi Orbitals
- Molecular Orbitals of The Allyl Cation, Allyl Radical, and Allyl Anion
- Pi Molecular Orbitals of Butadiene
- Reactions of Dienes: 1,2 and 1,4 Addition
- Thermodynamic and Kinetic Products
- More On 1,2 and 1,4 Additions To Dienes
- s-cis and s-trans
- The Diels-Alder Reaction
- Cyclic Dienes and Dienophiles in the Diels-Alder Reaction
- Stereochemistry of the Diels-Alder Reaction
- Exo vs Endo Products In The Diels Alder: How To Tell Them Apart
- HOMO and LUMO In the Diels Alder Reaction
- Why Are Endo vs Exo Products Favored in the Diels-Alder Reaction?
- Diels-Alder Reaction: Kinetic and Thermodynamic Control
- The Retro Diels-Alder Reaction
- The Intramolecular Diels Alder Reaction
- Regiochemistry In The Diels-Alder Reaction
- The Cope and Claisen Rearrangements
- Electrocyclic Reactions
- Electrocyclic Ring Opening And Closure (2) - Six (or Eight) Pi Electrons
- Diels Alder Practice Problems
- Molecular Orbital Theory Practice
18 Aromaticity
- Introduction To Aromaticity
- Rules For Aromaticity
- Huckel's Rule: What Does 4n+2 Mean?
- Aromatic, Non-Aromatic, or Antiaromatic? Some Practice Problems
- Antiaromatic Compounds and Antiaromaticity
- The Pi Molecular Orbitals of Benzene
- The Pi Molecular Orbitals of Cyclobutadiene
- Frost Circles
- Aromaticity Practice Quizzes
19 Reactions of Aromatic Molecules
- Electrophilic Aromatic Substitution: Introduction
- Activating and Deactivating Groups In Electrophilic Aromatic Substitution
- Electrophilic Aromatic Substitution - The Mechanism
- Ortho-, Para- and Meta- Directors in Electrophilic Aromatic Substitution
- Understanding Ortho, Para, and Meta Directors
- Why are halogens ortho- para- directors?
- Disubstituted Benzenes: The Strongest Electron-Donor "Wins"
- Electrophilic Aromatic Substitutions (1) - Halogenation of Benzene
- Electrophilic Aromatic Substitutions (2) - Nitration and Sulfonation
- EAS Reactions (3) - Friedel-Crafts Acylation and Friedel-Crafts Alkylation
- Intramolecular Friedel-Crafts Reactions
- Nucleophilic Aromatic Substitution (NAS)
- Nucleophilic Aromatic Substitution (2) - The Benzyne Mechanism
- Reactions on the "Benzylic" Carbon: Bromination And Oxidation
- The Wolff-Kishner, Clemmensen, And Other Carbonyl Reductions
- More Reactions on the Aromatic Sidechain: Reduction of Nitro Groups and the Baeyer Villiger
- Aromatic Synthesis (1) - "Order Of Operations"
- Synthesis of Benzene Derivatives (2) - Polarity Reversal
- Aromatic Synthesis (3) - Sulfonyl Blocking Groups
- Birch Reduction
- Synthesis (7): Reaction Map of Benzene and Related Aromatic Compounds
- Aromatic Reactions and Synthesis Practice
- Electrophilic Aromatic Substitution Practice Problems
20 Aldehydes and Ketones
- What's The Alpha Carbon In Carbonyl Compounds?
- Nucleophilic Addition To Carbonyls
- Aldehydes and Ketones: 14 Reactions With The Same Mechanism
- Sodium Borohydride (NaBH4) Reduction of Aldehydes and Ketones
- Grignard Reagents For Addition To Aldehydes and Ketones
- Wittig Reaction
- Hydrates, Hemiacetals, and Acetals
- Imines - Properties, Formation, Reactions, and Mechanisms
- All About Enamines
- Breaking Down Carbonyl Reaction Mechanisms: Reactions of Anionic Nucleophiles (Part 2)
- Aldehydes Ketones Reaction Practice
21 Carboxylic Acid Derivatives
- Nucleophilic Acyl Substitution (With Negatively Charged Nucleophiles)
- Addition-Elimination Mechanisms With Neutral Nucleophiles (Including Acid Catalysis)
- Basic Hydrolysis of Esters - Saponification
- Transesterification
- Proton Transfer
- Fischer Esterification - Carboxylic Acid to Ester Under Acidic Conditions
- Lithium Aluminum Hydride (LiAlH4) For Reduction of Carboxylic Acid Derivatives
- LiAlH[Ot-Bu]3 For The Reduction of Acid Halides To Aldehydes
- Di-isobutyl Aluminum Hydride (DIBAL) For The Partial Reduction of Esters and Nitriles
- Amide Hydrolysis
- Thionyl Chloride (SOCl2)
- Diazomethane (CH2N2)
- Carbonyl Chemistry: Learn Six Mechanisms For the Price Of One
- Making Music With Mechanisms (PADPED)
- Carboxylic Acid Derivatives Practice Questions
22 Enols and Enolates
- Keto-Enol Tautomerism
- Enolates - Formation, Stability, and Simple Reactions
- Kinetic Versus Thermodynamic Enolates
- Aldol Addition and Condensation Reactions
- Reactions of Enols - Acid-Catalyzed Aldol, Halogenation, and Mannich Reactions
- Claisen Condensation and Dieckmann Condensation
- Decarboxylation
- The Malonic Ester and Acetoacetic Ester Synthesis
- The Michael Addition Reaction and Conjugate Addition
- The Robinson Annulation
- Haloform Reaction
- The Hell–Volhard–Zelinsky Reaction
- Enols and Enolates Practice Quizzes
23 Amines
- The Amide Functional Group: Properties, Synthesis, and Nomenclature
- Basicity of Amines And pKaH
- 5 Key Basicity Trends of Amines
- The Mesomeric Effect And Aromatic Amines
- Nucleophilicity of Amines
- Alkylation of Amines (Sucks!)
- Reductive Amination
- The Gabriel Synthesis
- Some Reactions of Azides
- The Hofmann Elimination
- The Hofmann and Curtius Rearrangements
- The Cope Elimination
- Protecting Groups for Amines - Carbamates
- The Strecker Synthesis of Amino Acids
- Introduction to Peptide Synthesis
- Reactions of Diazonium Salts: Sandmeyer and Related Reactions
- Amine Practice Questions
24 Carbohydrates
- D and L Notation For Sugars
- Pyranoses and Furanoses: Ring-Chain Tautomerism In Sugars
- What is Mutarotation?
- Reducing Sugars
- The Big Damn Post Of Carbohydrate-Related Chemistry Definitions
- The Haworth Projection
- Converting a Fischer Projection To A Haworth (And Vice Versa)
- Reactions of Sugars: Glycosylation and Protection
- The Ruff Degradation and Kiliani-Fischer Synthesis
- Isoelectric Points of Amino Acids (and How To Calculate Them)
- Carbohydrates Practice
- Amino Acid Quizzes
25 Fun and Miscellaneous
- A Gallery of Some Interesting Molecules From Nature
- Screw Organic Chemistry, I'm Just Going To Write About Cats
- On Cats, Part 1: Conformations and Configurations
- On Cats, Part 2: Cat Line Diagrams
- On Cats, Part 4: Enantiocats
- On Cats, Part 6: Stereocenters
- Organic Chemistry Is Shit
- The Organic Chemistry Behind "The Pill"
- Maybe they should call them, "Formal Wins" ?
- Why Do Organic Chemists Use Kilocalories?
- The Principle of Least Effort
- Organic Chemistry GIFS - Resonance Forms
- Reproducibility In Organic Chemistry
- What Holds The Nucleus Together?
- How Reactions Are Like Music
- Organic Chemistry and the New MCAT
26 Organic Chemistry Tips and Tricks
- Common Mistakes: Formal Charges Can Mislead
- Partial Charges Give Clues About Electron Flow
- Draw The Ugly Version First
- Organic Chemistry Study Tips: Learn the Trends
- The 8 Types of Arrows In Organic Chemistry, Explained
- Top 10 Skills To Master Before An Organic Chemistry 2 Final
- Common Mistakes with Carbonyls: Carboxylic Acids... Are Acids!
- Planning Organic Synthesis With "Reaction Maps"
- Alkene Addition Pattern #1: The "Carbocation Pathway"
- Alkene Addition Pattern #2: The "Three-Membered Ring" Pathway
- Alkene Addition Pattern #3: The "Concerted" Pathway
- Number Your Carbons!
- The 4 Major Classes of Reactions in Org 1
- How (and why) electrons flow
- Grossman's Rule
- Three Exam Tips
- A 3-Step Method For Thinking Through Synthesis Problems
- Putting It Together
- Putting Diels-Alder Products in Perspective
- The Ups and Downs of Cyclohexanes
- The Most Annoying Exceptions in Org 1 (Part 1)
- The Most Annoying Exceptions in Org 1 (Part 2)
- The Marriage May Be Bad, But the Divorce Still Costs Money
- 9 Nomenclature Conventions To Know
- Nucleophile attacks Electrophile
27 Case Studies of Successful O-Chem Students
- Success Stories: How Corina Got The The "Hard" Professor - And Got An A+ Anyway
- How Helena Aced Organic Chemistry
- From a "Drop" To B+ in Org 2 – How A Hard Working Student Turned It Around
- How Serge Aced Organic Chemistry
- Success Stories: How Zach Aced Organic Chemistry 1
- Success Stories: How Kari Went From C– to B+
- How Esther Bounced Back From a "C" To Get A's In Organic Chemistry 1 And 2
- How Tyrell Got The Highest Grade In Her Organic Chemistry Course
- This Is Why Students Use Flashcards
- Success Stories: How Stu Aced Organic Chemistry
- How John Pulled Up His Organic Chemistry Exam Grades
- Success Stories: How Nathan Aced Organic Chemistry (Without It Taking Over His Life)
- How Chris Aced Org 1 and Org 2
- Interview: How Jay Got an A+ In Organic Chemistry
- How to Do Well in Organic Chemistry: One Student's Advice
- "America's Top TA" Shares His Secrets For Teaching O-Chem
- "Organic Chemistry Is Like..." - A Few Metaphors
- How To Do Well In Organic Chemistry: Advice From A Tutor
- Guest post: "I went from being afraid of tests to actually looking forward to them".
But diazomethane gives methyl ethers too in presence of hbf4 or bf3 can you share its mechanism as in what special does hbf4 or bf3 does such that reaction proceeds
Right. So the problem with doing it with normal alcohols is that alcohols are not acidic enough to protonate the CH2 on diazomethane. The solution is to use a protic acid like HBF4 as you mentioned. This gives CH3-N2(+) which is an excellent alkylating agent.
I think you stink
OK
need help reaction with propane and CH2N2
Under normal conditions, no reaction, but if the CH2N2 is agitated photochemically there could be formation of carbene and C-H insertion to either C-1 or C-2 of propane. Not something I would recommend.
In the late 1980s in the all the way into the mid- 1990s, as an Sr Laboratory Tech I set up the organic synthesis using Diazald® kit through Aldrich .
I perform 200 to 300 herbicide analyses per year through MTH 8150 SW 846. 3 version and I extracted my own samples.
I was well trained and well-versed in the safety hazards to being very cautious, and handling during the organic synthesis.
No ground glass joints!!. Ice down bath in the receiving flask. The reactions took place under a fume Hood and behind safety tempered blast shield.
Everything was smooth glass joints, triple rinse with petroleum ether production of least 100 to 200 milliliters of diazomethane at the end the synthesis was placed put into 4 or 5 40 mil Amber – Brown Teflon sealed capped then stored at 4 degrees Celsius in the laboratory refrigerator. Until was needed for reagent during the second part of the extraction process of MTH 8150.
After the rigorous extraction of at least 10 to 15 samples per day which took two days. Or 2/8 hour shift work., after the extraction of samples with diethyl ether was almost complete post Nordstrom blowdown throughs volume Zymark turbovap nitrogen blowdown. I would use the diazomethane 1 mil into each vial let it react for at least 2 to 3 minutes until bubbling stopped or I add silicic acid to stop the reaction. Transferred to GC vial be via pipette, capped, transferred to the GC /MS . For years, I never gave any thought about being injured on the job.
Great comment. Thank you. Were you converting carboxylic acids to esters? Are carboxylic acids present in a large majority of herbicides?
Why use this reagent when you can just use Fischer esterification?
Great question Eric. It depends on scale. For large scale reactions (say a gram or more) the Fischer is probably your best bet for reasons of cost and safety. It’s “set and forget”, come back in a few hours or the next morning and it will be done.
However a lot of exploratory organic chemistry is done on 50-100 mg of material (or less!) and in these cases, setting up a Fischer with a Dean-Stark trap isn’t very effective. Having a reagent available such as diazomethane that can quickly and easily (20 minutes !) install a methyl ester on a carboxylic acid is a huge time saver.
Thank you for creating not only the best organic chemistry website but really the only website that is of any help. I am taking organic chemistry II right now and this site is a lifesaver! It is a better resource than my textbook by far, from the amount of information to the way you explain everything. Just wanted to let you know I really appreciate your work!
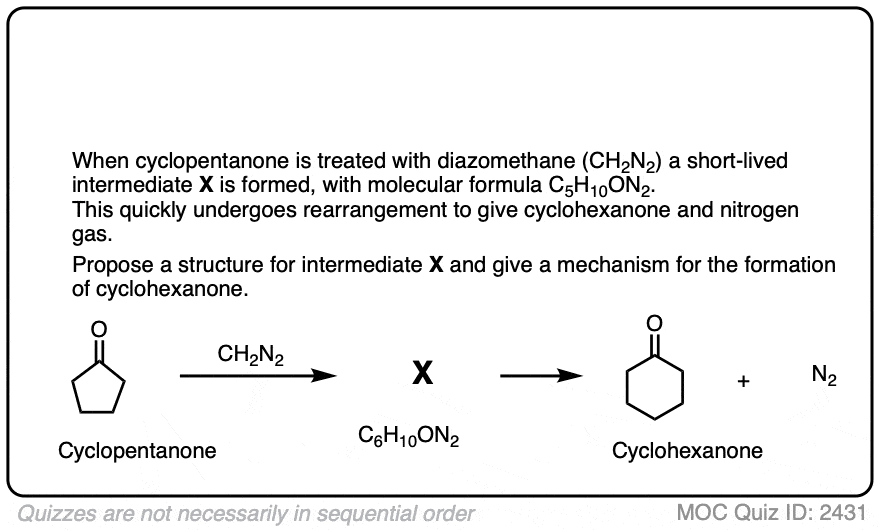
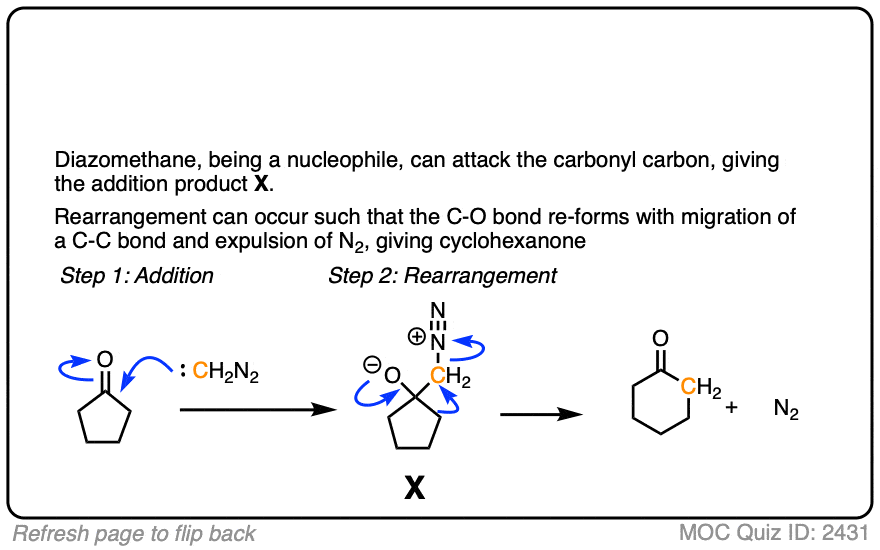
If we add this compound (diazomethane) to cyclopentanal, will we get cyclohexanal. If yes, is this type of reaction called kohler synthesis?
I’m not aware of the Kohler synthesis. What you’re describing is similar to the Buchner reaction: https://en.wikipedia.org/wiki/B%C3%BCchner%E2%80%93Curtius%E2%80%93Schlotterbeck_reaction
CH2N2 also reacts with alcohol (in presence of HBF4 ) right?
Yes, in the presence of strong acid, CH2N2 is protonated to CH3-N2(+) which is a superb alkylating agent. This will react with alcohols to give methyl ethers.
The new multiple-pesticide residue methods (MRM) were designated as NIOSH 5605 and 9204. They have not been published yet. In fact NIOSH hasn’t even reviewed them yet.(they had some cutbacks just after we finished the methods). We are in contact with them and sometime this month or next they want to talk to us about getting the final versions of the methods. So far 5602 and 9201 are the only methods we did that use diazomethane that have been published (can find them by searching “NIOSH 5602” or 9201 on line). Those published methods do not discuss the effects of diazomethane that we will report in 5605 and 9204. At this point the latter two methods are still NIOSH property and we are scheduled to work with NIOSH contacts later this month or next to get these method to them for final review. (It takes forever.)
These later NIOSH methods do not in themselves delve into the mechanism, it just reports the phenomenon as a caution to those using diazomethane in a MRM involving both acids and neutrals.
John M. Reynolds
James,
Loved your expose’. I have been using diazomethane for…I think 30-40 years (I’m 75, and still in one piece). Use it for pesticide analysis by GC-MS. Love it because it methylates sterically hindered benzoic acids, and phenols at the same time, AND does not affect my non-acid pesticides…so I can analyze them all together at once, when I want to. Discovered and reported that in NIOSH methods NMAM 9201 and 5602. In a method now being written up for NIOSH I evaluated the effects of diazomethane on dozens more of non-acid pesticides. It methylates imides (with an ionizable proton, e.g., acephate), and some keto-enol types (e.g., warfarin). It causes alpha-cyano pyrethroids to racemize around the benzylic carbon. Haven’t figured that one out. These effects are being reported in upcoming NIOSH methods. Couldn’t get it to methylate thio-acids. Have to resort to iodomethane.
John M. Reynolds
Fantastic comment. Very interesting. CH2N2 formation of methyl esters is definitely my favorite reaction to run: add, wait for bubbling to stop, then concentrate. Could you share a link to your methods?
Hi!
I have just find this website some minutes ago. I am a girl from Hungary, and last week I made an experiment with diazomethane, but I do not know, what happened, and I am really curious about your opinion.
So I wanted to make methyl chloroacetate, that’s why I solve 1 g chloroacetic acid in 10 ml THF and add diazomethane to that compound. I was waiting for 30 minutes and add 0,5 ml water. Then I evaporate the THF, and what I got was – in the beginning yellow, then really RED thing. I found it very strange, because I had thought the esther will be clear. My teacher check it in MS, and we did not have the molar mass of methyl chloroacetate in the chromatogram. Do you have any idea what happens? I am really excited!
Thank you very much in advance,
Veronika
Thank you very much for this invaluable support. Many android users we would also love to have thiss type of support on our cell phones, do you plan to relase the app also for android?. Thanks again!
does diazomethane react with alcohols, e.g n propanol ?
No, the first step is protonation, and alcohols are not acidic enough to perform this step.
I’ve been thinking about this for the past couple of days and did some reading up on diazomethane, but still haven’t been able to figure out why it has a tendency to explode when in the presence of high-surface-area materials or sharp edges. Does it have to do with the increased friction associated w/ high-surface areas and sharp edges?
I’m not 100% certain on this, but here’s my best guess. The surface of glass can be mildly acidic due to Si-OH groups. On a surface with enough hydrogen bond donors present you might be able to protonate/stabilize the CH2 and/or displace N2, and the exotherm in doing so may catalyze further reaction.
Ah yes, diazomethane. I had a friend in grad school, now graduated, who had to work with it once. He was legendary for his operational prowess in the lab—and diazomethane scared the pants off of him!
Notably, diazomethane is also probably the smallest ylide. I like to think of ylides as both nucleophilic and electrophilic at C—that’s why cyclopropanation works, because alkenes are both nucleophilic and electrophilic too (albeit on different atoms). Diazomethane, Simmons-Smith, Corey-Chaykovsky, and even rhodium carbenoids all operate on this principle.
I never thought of it as an ylide! but it makes perfect sense.
I was more scared by the making of it than actually using it, but it was definitely a two-hands-on-the-flask kind of reagent when you transfer it from the fridge to the hood.